Ester Expósito Age All Pictures And Videos #868
Unlock Now ester expósito age boutique media consumption. Pay-free subscription on our entertainment center. Dive in in a endless array of themed playlists exhibited in high definition, suited for deluxe watching supporters. With just-released media, you’ll always be informed. Discover ester expósito age chosen streaming in stunning resolution for a truly captivating experience. Sign up today with our community today to see restricted superior videos with without any fees, no sign-up needed. Look forward to constant updates and navigate a world of exclusive user-generated videos built for prime media fans. Grab your chance to see uncommon recordings—begin instant download! Access the best of ester expósito age specialized creator content with true-to-life colors and unique suggestions.
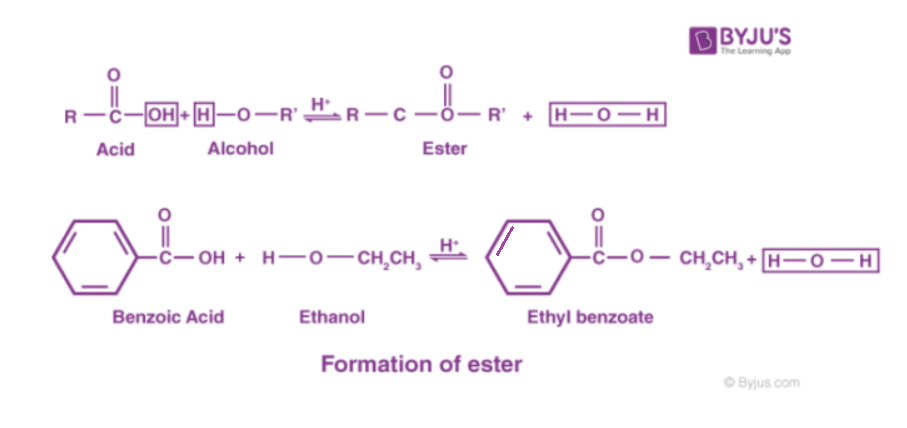
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (h) of at least one acidic hydroxyl group (−oh) of that acid is replaced by an organyl group (r ′) Ethyl acetate, for instance, is a commonly used solvent, and dialkyl phthalates are used as plasticizers to keep polymers from becoming brittle. [1] these compounds contain a distinctive functional group.
Ester Structural Formula
Ester, any of a class of organic compounds that react with water to produce alcohols and organic or inorganic acids The chemical industry uses esters for a variety of purposes Esters derived from carboxylic acids are the most common
- Josie Bennett Onlyfans
- Cece Rose Naked
- Kay Manuel Onlyfans Porn
- Angie Stylish Video Leak
- Very Old Grandma Naked
Learn about the different types and reactions of esters and more in this article.
The general structure of an ester is rcoor', where r and r' represent alkyl or aryl groups Esters are derived from the condensation reaction between a carboxylic acid and an alcohol, resulting in the elimination of water. An ester is an organic compound where the hydrogen in the compound's carboxyl group is replaced with a hydrocarbon group Esters are derived from carboxylic acids and (usually) alcohol.
Key takeaway an ester has an or group attached to the carbon atom of a carbonyl group. In this tutorial you will learn about the basic properties and structure of an ester functional group You will also learn about esterification and its mechanism. The meaning of ester is any of a class of often fragrant organic compounds that can be represented by the formula rcoor' and that are usually formed by the reaction between an acid and an alcohol with elimination of water.
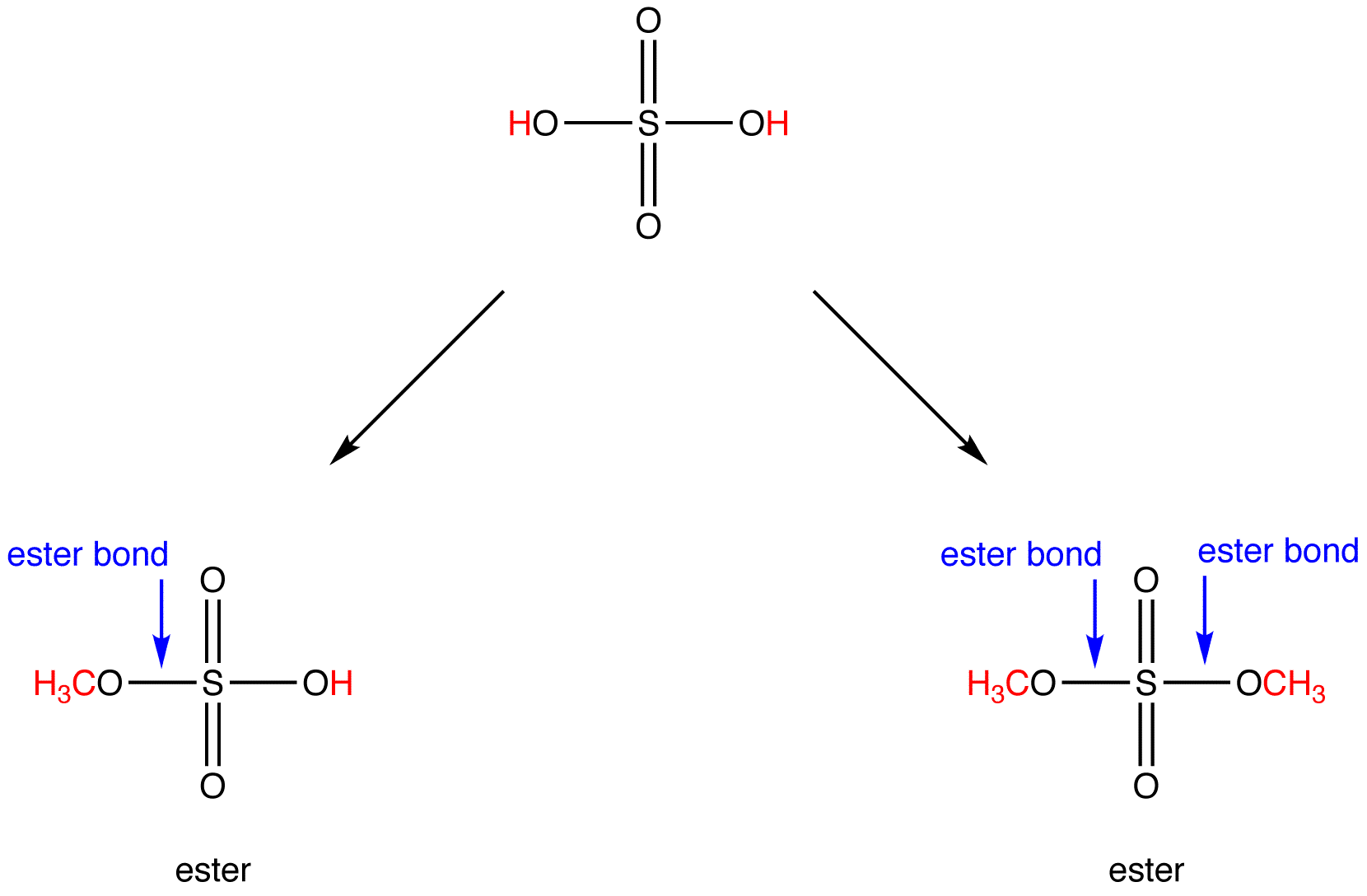
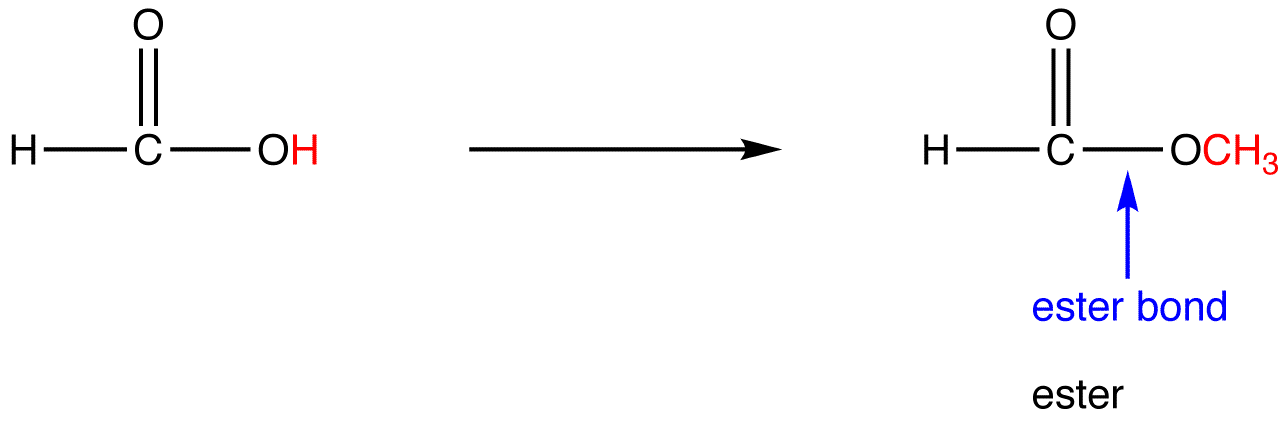
Illustrated glossary of organic chemistry ester
A molecule or functional group derived by condensation of an alcohol and an acid with simultaneous loss of water The most common type of ester is carboxylic ester (also called a carboxylate ester Frequently just called an ester), derived from a carboxylic acid. This could be an alkyl group like methyl or ethyl, or one containing a benzene ring like phenyl.
The ester linkage is also present in animal fats and in many biologically important molecules